 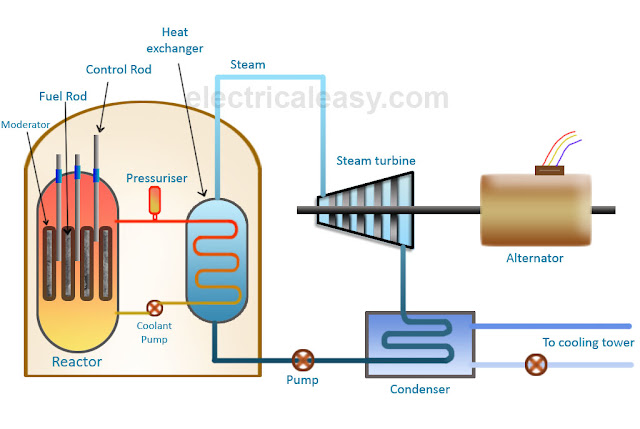
The rate of fission and power of a controlled chain reaction is constant, and can be controlled with the use of moderators and control rods. A controlled chain reaction allows for fission to occur in a self-regenerating manner while limiting the amount of energy produced to a sufficient but safe level. Controlled Chain ReactionĪ controlled chain reaction refers to a chain reaction whose rate of fission is controlled and thus the generated energy is also controlled. While moderators are used in uncontrolled chain reactions to increase the likelihood of successful fission, control rods are not used.  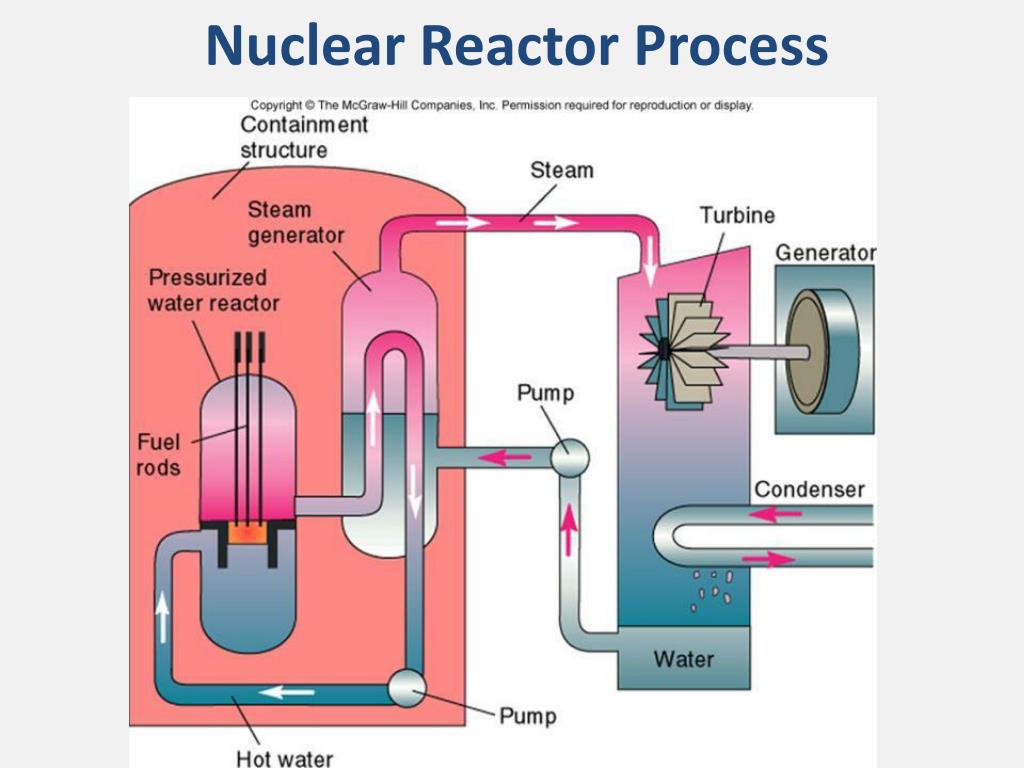
As a result, uncontrolled chain reactions are seen in the use of nuclear weapons. The rate of fission and power of an uncontrolled chain reaction are exponentially increasing. The amount of fissionable material required to facilitate this an uncontrolled chain reaction must exceed the critical mass. Uncontrolled chain reaction occurs when more than one further fission reaction is induced by neutrons emitted from fission. This kind of perfect reaction is called a critical reaction and the amount of fissionable material used to facilitate this one-for-one sustainability is called the critical mass (smallest amount of fissile material that will facilitate sustained chain reaction). In other words, one fission can induce another without any further intervention.Ĭritical reaction - each fission reaction produces exactly one further fission reaction and maintains constant reaction rate. Consequently, fission occurs to produce smaller nuclides that are more stable.Ī chain reaction in nuclear physics refers to the process during which nuclear fission reactions become self-sustaining. When the neutron is captured by the parent nucleus, an unstable nuclide is temporarily formed (due to changes in neutron to proton ratio). While a parent nuclide can undergo fission spontaneously, nuclear fission is sometimes induced by firing a neutron with sufficient kinetic energy into a heavy isotope (e.g. This process involves the formation of an intermediate (uranium-236) and emission of 3 neutrons. The emitted neutrons are important as they allow for chain reactions to occur.ĭiagram shows nuclear fission of uranium-235 to form two smaller nuclide fragments: krypton-92 and barium-141. In addition to small daughter nuclides, neutrons are sometimes formed from fission. By Einstein's mass-energy equivalence principle, this mass difference is transformed into energy. As a result, energy is released during nuclear fission, typically in the form of kinetic energy and heat.Ĭalculations involving nuclear fission can be found here. The total mass of products is also smaller than the mass of reactants. In nuclear fission, the binding energy of the products is greater than that of the parent nuclide. Nuclear fission is a type of transmutation where a large nuclide splits to form two smaller nuclides. Nuclear Fission, Controlled Chain Reactions and Nuclear Reactor What is Nuclear Fission? model and explain the process of nuclear fission, including the concepts of controlled and uncontrolled chain reactions, and account for the release of energy in the process (ACSPH033, ACSPH034).This topic is part of the HSC Physics course under the section Properties of the Nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
